When babies are born via C-section, they miss out on exposure to their mother's vaginal and fecal bacteria, which are crucial for developing a healthy gut microbiome. Instead, their microbiomes are shaped by bacteria from hospital environments and skin, often leading to imbalances. This can increase the risk of conditions like asthma, obesity, and allergies.
To address this, researchers are exploring ways to restore the microbiome in C-section infants. Key methods include:
- Vaginal Seeding: Swabbing the baby with the mother’s vaginal fluids after birth.
- Fecal Microbiota Transplantation (FMT): Administering maternal fecal bacteria to the baby.
- Synbiotic Interventions: Combining probiotics and prebiotics to promote gut health.
Studies show that these approaches can help normalize the gut microbiota, improve immune function, and reduce health risks. However, each method has its own challenges, and synbiotic solutions are emerging as a safer, more controlled option.
The first 100 days of life are critical for gut and immune development, making early interventions key to improving long-term health outcomes for C-section babies.
The New Birth Trend That’s All About Bacteria - IBD in the News
How C-Section Delivery Affects Infant Microbiomes
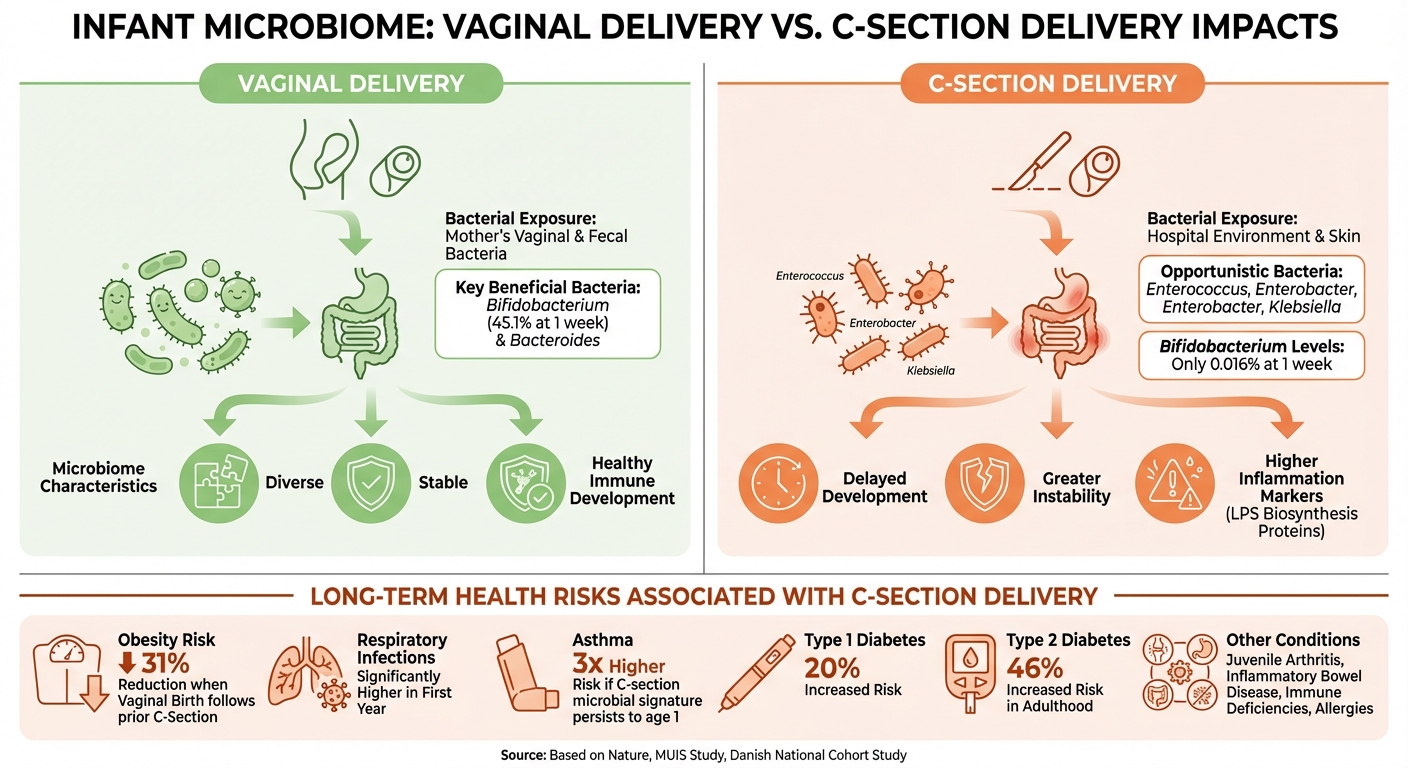
Vaginal vs C-Section Birth: Microbiome Differences and Health Impacts
Main Microbial Differences
The way a baby is delivered plays a key role in determining the first bacteria that populate their gut, shaping their health for years to come. Babies born vaginally are introduced to their mother's vaginal and fecal bacteria during delivery. This exposure helps establish a gut microbiome rich in beneficial bacteria such as Bifidobacterium and Bacteroides, which are crucial for developing a healthy immune system [5] [6].
On the other hand, babies born via C-section miss out on this exposure. Instead, their gut is initially colonized by bacteria from their mother's skin and the hospital environment [9] [11]. This often results in a microbiome dominated by opportunistic bacteria like Enterococcus, Enterobacter, and Klebsiella - microbes typically found on hospital surfaces rather than in a healthy gut [9] [11].
The difference is striking. One study found that, at just one week old, C-section babies had only 0.016% Bifidobacterium in their gut, compared to 45.1% in vaginally delivered infants [6]. Even breastfeeding, while beneficial, doesn’t completely close this gap. Data from the Microbiome Utrecht Infant Study (MUIS), which tracked 120 healthy Dutch infants between 2014 and 2016, showed that breastfed C-section babies still had significantly lower levels of Bifidobacterium compared to their vaginally delivered peers [6].
"Mode of delivery is a significant factor that affects the composition of the gut microbiota throughout the neonatal period, and into infancy." - Nature [11]
C-section babies also experience delayed gut microbiome development and greater instability in bacterial communities [5] [6]. Their microbiomes function differently too, with higher levels of lipopolysaccharide (LPS) biosynthesis proteins - linked to inflammation - being more common [5]. These changes in the microbiome set the stage for potential long-term health challenges.
Health Risks from Altered Microbiomes
The consequences of these microbial differences can be seen in real health outcomes. For instance, C-section delivery has been linked to an increased risk of obesity. Interestingly, if a mother delivers vaginally after a prior C-section, the younger child’s obesity risk drops by 31% [10]. The MUIS study also found that gut microbiota composition at just one week old could predict respiratory infections during the baby’s first year, with C-section infants experiencing significantly more infections [6].
"Birth by caesarean section (CS) has been associated with adverse effects on immune development, predisposing to infections, allergies, and inflammatory disorders." - Nature Communications [6]
The health risks don’t stop there. A Danish national cohort study of children born between 1977 and 2012 found that C-section delivery increased the likelihood of several immune-related conditions, including asthma, juvenile arthritis, inflammatory bowel disease, immune deficiencies, and even leukemia [9]. These risks persisted even after accounting for factors like maternal health and antibiotic use.
The problem extends to more serious concerns as well. A UK study analyzing 1,679 gut microbiota samples from 596 full-term babies revealed that C-section infants were more likely to harbor opportunistic pathogens. These pathogens often carried virulence factors and antimicrobial resistance genes, which could complicate treatment if infections occur [11]. These findings have inspired research into microbiome restoration techniques, which aim to address these imbalances through targeted interventions.
Clinical Research on Microbiome Restoration in C-Section Babies
Vaginal Seeding: Early Research
Since the mid-2010s, researchers have been investigating ways to restore the microbiome in babies born via C-section. One of the most studied techniques is vaginal seeding, also known as vaginal microbiota transfer (VMT). This process involves placing a sterile gauze in the mother's vagina for about an hour before the C-section. Shortly after birth - within 1 to 3 minutes - the gauze is used to swab the baby's mouth, face, and body [10] [12].
A study by Dominguez-Bello et al. demonstrated that vaginal seeding could partially restore the microbiome in these infants. Maternal vaginal fluids, which contain bacteria from the vagina, stool, skin, and oral cavity, serve as a diverse bacterial source for the newborn. In fact, 57.9% of the bacterial taxa in vaginal fluids overlap with those found in maternal stool [10] [2]. These early findings laid the groundwork for further research into the potential and limitations of this technique.
Recent Studies and Reviews
New studies have provided deeper insights into vaginal seeding. In June 2023, a double-blind, randomized, placebo-controlled trial involving 20 mother-infant pairs found that vaginal seeding significantly increased microbial transfer from mother to baby. The study also noted reduced alpha diversity in the newborns' skin (p = 0.047) and stool (p = 0.02). This pattern mirrors what is typically seen in babies delivered vaginally and exclusively breastfed, where beneficial microbes like Bifidobacterium dominate early microbial communities [10].
Another major study, published in Cell Host & Microbe in July 2023, involved 68 C-section infants in a triple-blind randomized controlled trial. It revealed that VMT not only sped up gut microbiota development but also improved neurodevelopmental outcomes at 6 months, as measured by the Ages and Stages Questionnaire (ASQ-3). The researchers concluded that VMT appears safe and may help normalize both gut microbiota and neurodevelopment in C-section babies [8].
However, not all studies have shown clear benefits. The ECOBABe trial, published in June 2021, tested an alternative method: administering 3 ml of maternal vaginal microbes orally to 25 C-section infants. This approach did not lead to significant differences in gut microbiome composition or function at 1 or 3 months [13].
Alternative Microbial Restoration Methods
Beyond vaginal seeding, other strategies have been explored. A proof-of-concept study conducted in October 2020 (NCT03568734) tested fecal microbiota transplantation (FMT) in 7 C-section-born infants. In this method, maternal fecal microbiota was orally administered to the infants. Within three months, the treated babies' gut microbiota closely resembled that of vaginally delivered infants. However, this approach requires rigorous maternal pathogen screening to ensure safety [1] [13].
Targeted synbiotic interventions have also shown promise. In an open-label randomized controlled trial by Tiny Health, 54 C-section-born infants participated in a personalized microbiome program that included longitudinal testing, coaching, and tailored synbiotic treatments. Published in Pediatric Allergy and Immunology in early 2025, the study found that these interventions significantly increased Bifidobacterium levels (p = 0.025) and reduced the prevalence of atopic dermatitis (p = 0.031), with an odds ratio of 0.17 (95% CI, 0.023–0.723) [4]. These results highlight the potential of personalized approaches to address microbiome-related concerns in C-section infants.
sbb-itb-1bbfe7f
Immune and Developmental Outcomes of Microbiome Restoration
Short-Term Benefits
The early months of life are pivotal for the gut microbiome's role in teaching the immune system to differentiate between harmful pathogens and harmless substances [14][7]. For babies born via C-section, restoring their microbiome early on can lead to noticeable improvements in immune function and developmental progress. One compelling example comes from a July 2023 triple-blind randomized controlled trial involving 68 cesarean-delivered infants. This study found that applying Vaginal Microbiota Transfer (VMT) immediately after birth resulted in significantly higher neurodevelopmental scores on the Ages and Stages Questionnaire (ASQ-3) at 6 months. Additionally, within just 42 days, VMT was shown to regulate key metabolic pathways, including those for carbohydrate and amino acid metabolism [8].
"VMT is associated with improved neurodevelopment in cesarean-born infants." - Cell Host & Microbe [8]
Restoration efforts also help normalize the Th1/Th2 cell balance, which is often skewed toward a Th2 allergic response in C-section babies. These interventions increase essential immune markers like serum IgG and IL-12p70 levels while reducing colonization by opportunistic pathogens such as Klebsiella and Enterococcus [7][15]. Notably, infants with higher levels of Bifidobacterium just one week after birth experienced significantly fewer respiratory infections during their first year (0–2 events compared to 3–7 events) [15].
Further evidence comes from a 2025 trial involving 54 C-section infants. Those who received personalized microbiome coaching showed significantly higher Bifidobacterium levels (p = 0.025) and a reduced odds ratio for atopic dermatitis (0.17; 95% CI, 0.023–0.723) compared to controls [4]. These findings underscore the potential of targeted microbiome interventions to improve early immune and developmental outcomes, laying the groundwork for longer-term health benefits.
Long-Term Immune Health Effects
The benefits of microbiome restoration extend beyond infancy, potentially reducing the risk of chronic immune-related conditions. Studies show that children who retain a C-section microbial signature at age 1 are three times more likely to develop asthma by age 6 [16]. However, if the gut microbiota recovers and matures normally by their first birthday, this elevated risk can be significantly diminished [17].
"Even though a child is born by caesarean section and has an immense early microbial perturbation, this may not lead to a higher risk of asthma if the microbiome matures sufficiently before age 1 year." - Jakob Stokholm, Senior Scientist, The Copenhagen Prospective Study on Asthma in Childhood [17]
Microbiome restoration may also help interrupt the "atopic march", a progression that starts with atopic dermatitis in infancy and can lead to food allergies, allergic rhinitis, and asthma later in childhood [16][18]. Additionally, C-section delivery has been linked to a 20% higher risk of childhood-onset Type 1 diabetes and a 46% greater risk of developing Type 2 diabetes in adulthood compared to vaginal delivery [18]. Early microbiome interventions appear to support normal energy metabolism and promote immune tolerance, potentially lowering the risk of these metabolic and autoimmune conditions [18].
While more long-term randomized clinical trials are needed to confirm whether early microbiome restoration can definitively prevent chronic diseases like asthma, obesity, and Type 1 diabetes, current evidence highlights the critical importance of the first 100 days to one year of life for immune system development [17][18]. This period represents a key window for setting the stage for lifelong health.
Synbiotic Interventions for Microbiome Reset
What Are Synbiotics?
Synbiotics combine probiotics (live, beneficial microorganisms) with prebiotics (nutrients that help those microorganisms thrive), offering a dual-action approach to support gut health [20]. This combination is particularly useful for infants born via C-section, as they miss out on the natural transfer of maternal bacteria during birth.
C-section deliveries disrupt the natural colonization process, delaying the growth of Bifidobacterium and reducing Bacteroidota species in the infant gut [19]. While prebiotics alone can nourish existing gut bacteria, they cannot introduce missing strains. Synbiotics step in to fill this gap by providing both the beneficial bacteria and the nutrients they need to grow [20].
Clinical Evidence for Synbiotic Effectiveness
Research consistently shows that synbiotics are more effective than prebiotics alone for restoring gut health in C-section infants. For example, a multicenter trial (NCT03520764) conducted with healthy Chinese infants tested a synbiotic formula containing Bifidobacterium breve M-16V and scGOS/lcFOS. The findings revealed that the synbiotic restored Parabacteroides levels by 17 weeks and Bacteroides levels by 12 months, aligning them with levels seen in vaginally born infants [19].
"The synbiotic was confirmed to support the restoration of important gut colonizers in infants born by C-section with effects observed even beyond the intervention period."
– Wang Y. et al., European Journal of Clinical Nutrition [19]
Another randomized controlled trial (NCT06424691) conducted in early 2025 involved 54 full-term C-section infants. This study demonstrated that personalized synbiotic interventions significantly boosted Bifidobacterium levels (p = 0.025) and reduced the risk of atopic dermatitis by 83% (odds ratio of 0.17; 95% CI, 0.023–0.723) [4].
"A personalized microbiome-based program can modulate the gut microbiome of C-section–born infants and may reduce the risk of atopic conditions."
– Tiny Health Research Team, Pediatric Allergy and Immunology [4]
These interventions also lowered the "C-section index" to levels comparable to vaginal births and enhanced the abundance of functional genes involved in human milk oligosaccharide (HMO) digestion, like α-L-fucosidase (p = 0.019), which is vital for infant nutrition [4]. Such findings highlight the potential of targeted products like Begin Rebirth RE-1™ to accelerate microbiome recovery.
Begin Rebirth RE-1™: A Rapid Microbiome Reset
Building on this research, Begin Rebirth RE-1™ offers a targeted synbiotic solution designed specifically for C-section infants. This 3-in-1 formula combines prebiotics, probiotics, and postbiotics to restore gut balance and support the development of the gastrointestinal immune system [21]. Its formulation addresses microbial deficits commonly seen in C-section infants, promoting bacterial diversity and immune health.
The product utilizes Human Origin Strains (HOSt™), chosen for their ability to colonize the infant gut and support early immune development [4][21]. Each sachet delivers 500 billion CFU and 4.5 g of fiber (GOS and inulin) using the Lyosublime™ delivery system, which preserves CFU viability and ensures effective absorption without refrigeration. Available in 7-day, 4-week, and 12-week programs, Begin Rebirth RE-1™ offers flexible options for microbiome restoration.
Initiating synbiotic interventions like Begin Rebirth RE-1™ within the first three months of life is critical, as this period represents a key window for microbiome recovery in C-section infants [21]. When combined with breastfeeding, the benefits are even greater, as natural milk oligosaccharides work synergistically with the synbiotic formula to enhance gut health and immune function.
Conclusion
C-section deliveries can interfere with the natural transfer of maternal bacteria, leading to a reduced presence of microbes that play a crucial role in supporting immune and metabolic health [3]. This disruption in the microbiome has been linked to increased risks of conditions like asthma, atopic dermatitis, Type 1 diabetes, and childhood obesity, highlighting the importance of early microbiome restoration [3][22].
While vaginal seeding has gained attention as a way to partially address this issue, it comes with notable safety concerns. By exposing newborns to maternal vaginal fluids, this method aims to "naturalize" the baby's microbial development during the first year of life [2]. However, risks like the potential transmission of pathogens, including Group B streptococcus, and its limited ability to fully restore gut-adapted bacterial families raise questions about its widespread use. As Dr. Allan Walker of Harvard Medical School notes, current data do not yet support routine implementation of microbial restoration techniques [23].
A safer and more controlled alternative lies in advanced synbiotic solutions. Products like Begin Rebirth RE-1™ combine prebiotics, probiotics, and postbiotics with Human Origin Strains (HOSt™) to help rebuild gut health without the risks associated with raw maternal transfers. When paired with breastfeeding, these interventions have been shown to significantly increase beneficial bacteria levels and reduce the likelihood of atopic conditions by as much as 83% [4].
Parents and caregivers should work closely with healthcare providers to explore the best microbiome restoration options tailored to their needs. Whether through carefully supervised vaginal seeding or advanced synbiotic approaches, taking steps to support gut health early on can help reduce the long-term health risks tied to C-section deliveries.
FAQs
How does being born via C-section affect a baby’s microbiome?
Babies born via C-section encounter a different microbial introduction compared to those delivered vaginally. Instead of receiving microbes from their mother’s vaginal microbiota, they are mainly exposed to microbes from the skin and the hospital environment. This shift can slow the development of a diverse and balanced gut microbiome, which plays a key role in supporting immune function and metabolism.
Studies suggest that C-section deliveries may lead to slower microbial colonization and lower microbiome diversity in infants. This altered start could affect immune system development and potentially raise the likelihood of certain health issues later in life. To address this, researchers are investigating ways to restore the gut microbiome in C-section babies, aiming to support their long-term health.
What health risks can C-section babies face due to an altered microbiome?
Babies delivered by C-section may encounter certain health challenges tied to differences in their gut microbiome. Unlike vaginal births, C-sections bypass the natural transfer of beneficial maternal bacteria, which leaves these infants with a gut microbiome that often contains more microbes typically found in hospital settings. This imbalance can interfere with gut health and has been linked to a higher likelihood of issues such as respiratory infections, asthma, allergies, and even obesity.
The absence of early exposure to maternal vaginal bacteria might also impact the development of the immune system, potentially making these babies more prone to immune-related conditions. Research suggests that techniques aimed at restoring the microbiome could help promote a healthier microbial balance in C-section infants.
Are synbiotics safe and effective for restoring the microbiome in C-section babies?
Yes, synbiotics - combinations of prebiotics and probiotics - are a safe and effective option for helping restore the gut microbiome in babies born via C-section. Studies show that synbiotics encourage the growth of beneficial bacteria like Bifidobacteria, which tend to be lower in C-section infants. This can help balance their gut microbiome and support the development of a healthy immune system early on.
Research also highlights that synbiotic treatments can address microbiota imbalances caused by C-section deliveries or antibiotic use. These treatments are particularly effective in breastfed babies, as they work in tandem with breast milk to create a supportive gut environment. Synbiotics provide a practical way to counteract early disruptions in the microbiome, offering important benefits for C-section infants.