Postbiotics, non-living microbial products, are gaining attention for their role in immune health. Unlike probiotics (live microbes) or prebiotics (microbial food), postbiotics consist of inactive microbes and their components, such as cell walls, peptides, and fermentation byproducts like short-chain fatty acids (SCFAs). Their stability, safety, and ability to directly interact with the immune system make them a promising tool for improving gut and immune health.
Key Takeaways:
- How They Work: Postbiotics interact with immune receptors (e.g., Toll-like receptors) to balance pro- and anti-inflammatory responses, strengthen gut barriers, and enhance mucosal defenses.
- Advantages: They are stable, require no refrigeration, and are safe for all age groups, including newborns and immunocompromised individuals.
- Research Highlights: Clinical trials show benefits like reduced infections, improved gut health markers (e.g., secretory IgA), and fewer inflammatory conditions.
- Applications: Postbiotics are used in immune support, gut health products, and synbiotic formulations (combined with probiotics and prebiotics).
Postbiotics offer a practical, safe, and effective option for supporting immune health, but further research is needed to understand their full potential and optimize their use in personalized healthcare.
Episode 18: The definition of postbiotics
How Postbiotics Modulate the Immune System
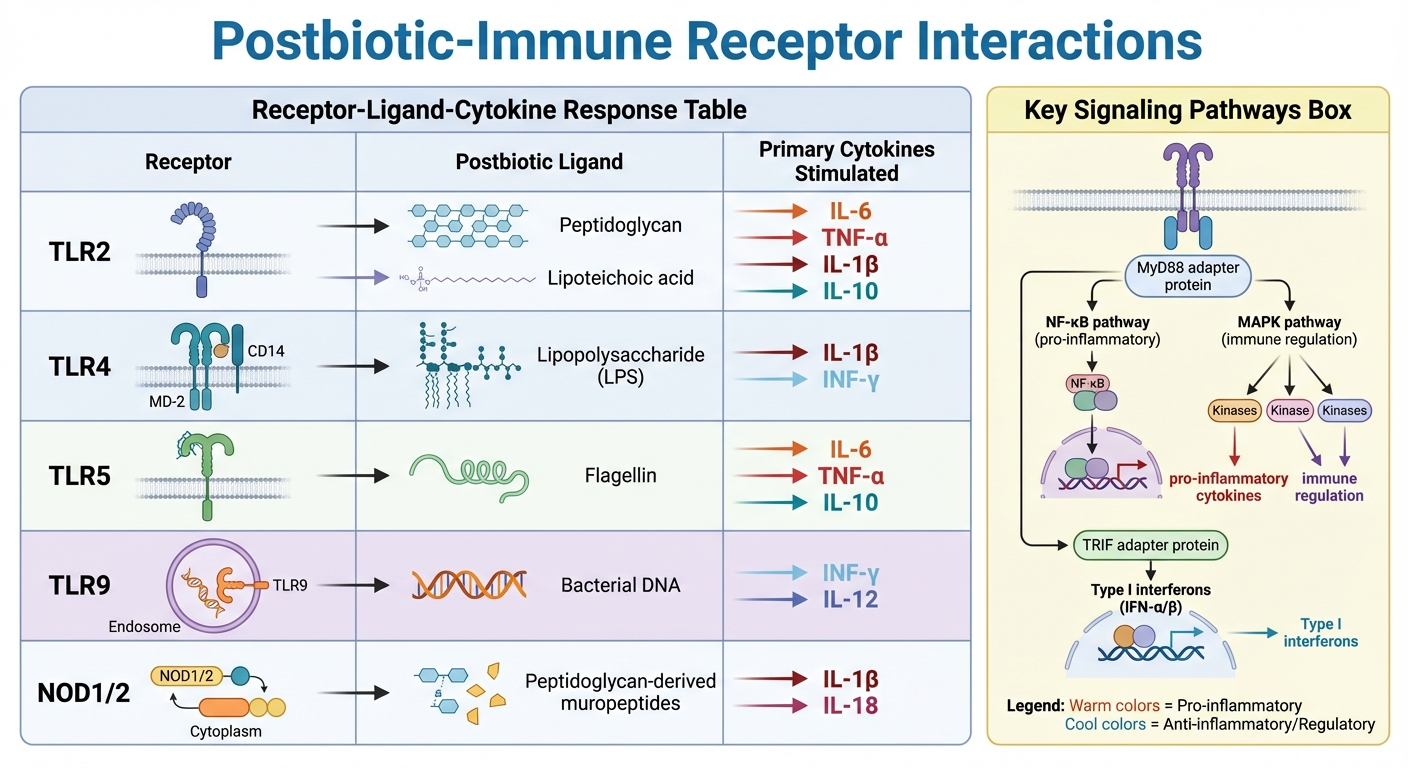
How Postbiotics Interact with Immune Receptors and Trigger Cytokine Responses
Interaction with Pattern Recognition Receptors
Postbiotics engage with the immune system by interacting with Pattern Recognition Receptors (PRRs), which act as immune system sensors. These receptors include Toll-like receptors (TLRs) and Nucleotide-binding Oligomerization Domain (NOD)-like receptors. Each postbiotic binds to specific receptors, influencing immune responses [3][4].
For example:
- Peptidoglycan and muropeptides, components of bacterial cell walls, bind to TLR2, NOD1, and NOD2, regulating inflammatory signaling [3][9][4].
- Lipoteichoic acids (LTA) from Gram-positive bacteria activate TLR2 and TLR6, driving the production of cytokines like IL-6 and TNF-α [3][2].
- Lipopolysaccharides (LPS) from Gram-negative bacteria are recognized by the TLR4/CD14 complex. While they can trigger inflammatory responses, postbiotics may modify this interaction to promote tolerance [3][4].
- Flagellin, a protein in bacterial flagella, interacts with TLR5 to activate specific cytokine pathways [3][4].
When these receptors are activated, they initiate intracellular signaling, notably through the NF-κB and MAPK pathways. These pathways regulate the production of cytokines and antimicrobial molecules [3][10]. Additionally, postbiotics influence signaling adapter proteins like MyD88 and TRIF. MyD88 primarily drives pro-inflammatory cytokine production, while TRIF promotes the release of Type I interferons (IFN-α/β) [3].
| Receptor | Postbiotic Ligand | Primary Cytokines Stimulated |
|---|---|---|
| TLR2 | Peptidoglycan, Lipoteichoic acid | IL-6, TNF-α, IL-1β, IL-10 |
| TLR4 | Lipopolysaccharide (LPS) | IL-1β, INF-γ |
| TLR5 | Flagellin | IL-6, TNF-α, IL-10 |
| TLR9 | Bacterial DNA | INF-γ, IL-12 |
| NOD1/2 | Peptidoglycan-derived muropeptides | IL-1β, IL-18 |
Through these interactions, postbiotics fine-tune cytokine production, ensuring a balanced immune response.
Cytokine Network Modulation
Postbiotics play a critical role in balancing pro-inflammatory cytokines (like IL-6, TNF-α, and IL-1β) with anti-inflammatory ones (such as IL-10 and TGF-β) [6][4]. This equilibrium is essential for maintaining immune homeostasis, allowing the body to fight infections while avoiding excessive inflammation.
One way postbiotics achieve this is by suppressing NF-κB translocation, which helps reduce chronic inflammation [6]. Short-chain fatty acids (SCFAs), a key group of postbiotics, promote the differentiation of regulatory T cells (Tregs). These Tregs release IL-10, a cytokine known for its anti-inflammatory properties, helping to maintain immune balance [6][4].
Interestingly, postbiotics can also stimulate the immune system when needed. For instance, they promote IL-12 production, which activates natural killer (NK) and Th1 cells, enhancing the body's ability to combat pathogens and tumors [4][3]. This dual role - calming inflammation while bolstering defenses - highlights their importance in immune regulation.
These cytokine shifts also strengthen the gut's protective barriers.
Strengthening the Gut Barrier and Mucosal Defense
Postbiotics support gut health by reinforcing the intestinal barrier through several mechanisms. Exopolysaccharides (EPS), for instance, enhance the expression of intercellular adhesion proteins like ZO-1 and occludin via STAT3 activation [11]. They also reduce the expression of Claudin-2, a protein linked to increased gut permeability and dysfunction [11].
Additionally, postbiotics stimulate the production of mucin glycoproteins, which form a protective layer over the gut lining. These components also prevent pathogenic biofilm formation and compete with harmful microbes for colonization sites [7]. Specific proteins like p75 and p40, derived from Lactobacillus rhamnosus, protect the gut lining by reducing epithelial cell apoptosis [2].
Another critical function of postbiotics is boosting the production of secretory IgA (sIgA), the primary immunoglobulin in mucosal secretions. The Gut-Associated Lymphoid Tissue (GALT) produces about 80% of all immunoglobulins, with sIgA playing a key role in capturing antigens and preventing their penetration into mucous membranes [4]. Research on infants fed formulas containing postbiotic-producing strains (B. infantis R0033, B. bifidum R0071) showed increased fecal sIgA levels, suggesting enhanced mucosal immunity [10].
Finally, short-chain fatty acids (SCFAs), particularly butyrate, serve as a vital energy source for colonic epithelial cells (colonocytes). This supports villi growth and crypt development, further strengthening the gut barrier [11].
Key Findings from Recent Studies
In Vitro and Animal Studies
Laboratory studies have shed light on how postbiotics interact with the immune system at a molecular level. For instance, in cell cultures, postbiotics engage with Toll-like receptors (TLR2, TLR4, TLR9) found on monocytes, macrophages, and B lymphocytes. This interaction lowers levels of pro-inflammatory cytokines like TNF-α, IL-6, and IL-1β, while boosting anti-inflammatory mediators such as IL-10 and TGF-β [3][13].
Animal models provide further insights. In one study, protein-deprived mice treated nasally with peptidoglycan from L. rhamnosus CRL1505 (PG05) showed faster recovery of bone marrow myeloid cells and activated alveolar macrophages. The Laboratory of Immunobiotechnology emphasized:
"the key cellular component for the effects on mucosal immunity, which are unique and cannot be extrapolated to other L. rhamnosus or probiotic strains" [8].
In a SHIME® gut model, the yeast postbiotic EpiCor increased butyrate production in the proximal colon by 36.7% [12]. Meanwhile, exopolysaccharides from L. paraplantarum BGCG11 reduced IL-1β and TNF-α levels in animal models, showing potential to alleviate hyperalgesia [2]. Additionally, cell-free supernatants from L. acidophilus triggered apoptosis in HT-29 colon cancer cells and lowered TNF-α levels [13][7].
These findings from lab and animal research have set the stage for testing postbiotics in human trials.
Human Clinical Trials
Clinical trials have confirmed many of the findings observed in laboratory and animal studies. A 2024 study by Danone Nutricia examined the effects of a "Lactofidus" fermented formula on 432 healthy infants (0 to 28 days old). Results showed higher secretory IgA levels, reduced Clostridium difficile counts, and a marked decrease in infant colitis [1].
The INNOVA study assessed Nutribén® Innova 1 infant formula containing heat-inactivated Bifidobacterium animalis subsp. lactis (BPL1™ HT). Infants consuming this formula experienced lower rates of atopic dermatitis, bronchitis, and bronchiolitis compared to those on standard formulas [1]. Another trial involving heat-inactivated L. paracasei CBA L74 in young children found fewer episodes of diarrhea, pharyngitis, and laryngitis, alongside increased secretory IgA levels [1]. Similarly, heat-killed Lactobacillus acidophilus LB was shown to shorten the duration of diarrhea [1]. In adults, bacterial lysates derived from Bifidobacterium longum improved reactive skin conditions by modulating inflammatory markers [2].
These clinical results highlight the potential of postbiotics for inclusion in synbiotic formulations to enhance immune function.
Comparative Overview of Study Evidence
A combined analysis of in vitro, animal, and human studies underscores the consistent immune benefits of postbiotics.
| Study Type | Focus Area | Key Immune Outcomes | Limitations |
|---|---|---|---|
| In Vitro | Cell signaling & receptors | TLR activation, NF-κB inhibition, cytokine modulation (e.g., IL-10, TNF-α) | Lacks insights into host pharmacokinetics/dynamics |
| Animal | Systemic physiology | Bone marrow recovery, thymus atrophy reversal, enhanced disease resistance | Results may not directly apply to humans |
| Human Clinical | Clinical efficacy | Reduced infection risk, improved gut markers like SIgA, allergy mitigation | High cost; requires larger-scale studies |
Laboratory studies are particularly effective at uncovering mechanisms, such as enhancing tight junction proteins like zonula occludens-1. In contrast, human trials focus on measurable health outcomes, including shorter episodes of acute diarrhea and fewer respiratory infections [1]. Animal research often reveals effects that are harder to observe in humans, such as reversing thymus atrophy in malnourished models [8]. Pediatric trials have confirmed lab findings, with fermented formulas increasing fecal secretory IgA levels in infants [1].
One area needing further exploration is oncology. While lab studies suggest postbiotics can induce apoptosis in cancer cells, human evidence remains limited to their potential as adjunct therapies rather than standalone treatments [13][7]. Importantly, clinical trials have also verified the safety of postbiotics for use in immunocompromised individuals [2][7].
Altogether, these findings highlight the immune-supportive potential of postbiotics and offer valuable direction for future research.
sbb-itb-1bbfe7f
Practical Applications of Postbiotics for Immune Health
Potential Uses in Immune Support
Postbiotics are stepping into the spotlight as practical tools for supporting immune health. For instance, they help restore gut balance after antibiotic use and can ease symptoms of conditions like IBS and IBD by strengthening the gut lining and calming local inflammation [6]. Since the gut plays a central role in overall immunity, improving its health can have far-reaching effects on the body’s defense systems.
What sets postbiotics apart is their safety, making them a great option for groups that need extra care. Immunocompromised individuals, premature infants, pregnant women, and older adults can all benefit from postbiotic supplementation [2]. Veilumuthu Pattapulavar from the Vellore Institute of Technology highlights their promise:
"Postbiotics have the potential to serve as safe next-generation therapeutics without the need to administer live bacteria" [5].
This combination of safety and effectiveness positions postbiotics as key players in advanced synbiotic products.
Postbiotics in Synbiotic Formulations
One of the standout features of postbiotics is their stability. They remain effective during processing, storage, and even the journey through the digestive system [6]. Unlike probiotics, they don’t require refrigeration and can handle temperature changes, making them a practical choice for everyday use.
In 3-in-1 synbiotic formulations, postbiotics team up with prebiotics (which feed good bacteria) and probiotics (live bacteria that can shift microbiome composition) to offer a complete approach to gut health. While probiotics work over time to adjust the microbiome, postbiotics deliver immediate bioactive compounds that support the immune system and gut lining. The International Scientific Association of Probiotics and Prebiotics (ISAPP) defines postbiotics as "preparations of inanimate microorganisms and/or their components that confer a health benefit on the host" [6].
Begin Rebirth RE-1™: A Synbiotic Example
A real-world example of this synergy is Begin Rebirth RE-1™, a 3-in-1 synbiotic designed for a quick microbiome reset. Each sachet offers 500 billion CFU of probiotics, 4.5 g of prebiotic fiber (GOS and Inulin), and postbiotics, all delivered through the proprietary Lyosublime™ system. These formulations use Human Origin Strains (HOSt™) and are available in 7-day, 4-week, and 12-week programs, providing a versatile solution for gut and immune health - all without needing refrigeration.
This integrated approach reflects the growing trend of combining prebiotics, probiotics, and postbiotics into one easy-to-use product. It highlights how postbiotics, with their stability and immediate benefits, complement the longer-term effects of probiotics and the nourishing power of prebiotics. Together, they offer a comprehensive solution for gut health and immune support.
Research Gaps and Future Directions
Mechanistic Gaps in Immune Modulation
Although postbiotics hold great promise, there are still many unanswered questions that need to be tackled before they can be fully optimized for clinical use. One of the biggest challenges is that the molecular mechanisms behind postbiotics remain largely unknown. These mixtures of metabolites, cell fragments, and secreted factors are incredibly complex, and a staggering 80–90% of microbial metabolites are still unidentified. This lack of identification makes it tough to pinpoint how exactly postbiotics influence immune responses [14][15][16]. Bing Liang and Dongming Xing from The Affiliated Hospital of Qingdao University highlight this issue:
"The exact mechanism of action by which postbiotics exert specific effects has not been fully elucidated." [15]
Another hurdle is linking these metabolites to the specific gene networks they regulate within the host [16]. Without this connection, predicting how postbiotics interact with immune receptors like Toll-like receptors (TLRs) and nucleotide-binding oligomeric domain receptors (NODs) becomes nearly impossible [15][6]. Solving these mechanistic puzzles is critical for designing effective clinical trials that can deliver consistent and meaningful results.
Priorities for Clinical Trials
To truly understand the potential of postbiotics, larger, well-structured human trials involving diverse populations are urgently needed [18][5]. Much of the current evidence comes from either small-scale studies or animal research, which limits its applicability. For example, one recent trial involving stroke patients showed promising results, with reduced pneumonia rates and lower levels of inflammatory markers following postbiotic supplementation [19].
However, significant gaps remain in areas like chronic inflammatory skin conditions (e.g., psoriasis, rosacea, atopic dermatitis), food allergies, and metabolic disorders such as Type 2 diabetes [20][18][6]. Another issue is the lack of consistency in production methods, including how postbiotics are inactivated. This variability leads to inconsistent findings across different studies [14][17]. Future clinical trials need to standardize these methods and also focus on long-term safety monitoring to establish postbiotics as dependable therapeutic options.
Personalized Postbiotic Strategies
Because individual responses to postbiotics can vary greatly, tailoring interventions to each person's microbiome is essential [16][17]. Factors like baseline microbiome composition, diet, age, and medications all influence how a person might respond. This is where "metabotyping" comes in - analyzing how someone's unique gut flora processes compounds and produces metabolites [23]. By identifying these metabolic gaps, researchers can design targeted interventions.
For instance, if someone’s gut lacks the ability to convert polyphenols into urolithin A (a beneficial compound), specific supplementation could help bridge that gap [23]. As one study explains:
"It is not the core or unique key species of microbes that influence health or disease states. Instead, it is the conserved functional redundancies of microbial species that significantly vary among individuals." [16]
To make these personalized approaches more effective, clinicians should analyze patients' microbiomes before recommending postbiotics. This ensures that treatments complement existing gut bacteria rather than disrupt them. Additionally, metabolomic testing can help determine the right dosage for each individual [21][23]. This strategy is especially critical for vulnerable groups, such as newborns, whose developing immune systems are more receptive to targeted interventions [22]. By focusing on personalization, postbiotics can become a more precise and effective tool for supporting immune health.
Conclusion
Postbiotics are making waves in the field of immune health research. These non-living microbial products interact directly with immune receptors, influence cytokine activity, and fortify the gut barrier. They provide a reliable and safe alternative to live probiotics. As Sunita Aggarwal from the University of Delhi explains:
"Postbiotics are superior to probiotics because of their purity, ease of preparation, long shelf life, mass production capability, precise action and more targeted responses" [2].
Their potential is already being realized in practical applications. Since postbiotics don’t involve live bacteria, they sidestep safety concerns, making them especially suitable for vulnerable groups like immunocompromised individuals, premature infants, and older adults. Products like Begin Rebirth RE-1™, which combine prebiotics, probiotics, and postbiotics, are paving the way for comprehensive gut and immune support.
However, there’s still work to be done. Larger clinical studies involving diverse groups are necessary to refine dosing guidelines and better understand how individual responses vary. The future of postbiotics likely lies in personalized medicine, where interventions are tailored to each person’s unique microbial makeup.
Despite these challenges, the potential is undeniable. Postbiotics address the stability and viability issues of live probiotics while delivering targeted immune benefits. As John Godwin Christopher from the Vellore Institute of Technology points out:
"Postbiotics have the potential to serve as safe next-generation therapeutics without the need to administer live bacteria" [5].
Ongoing research into their mechanisms and personalized applications will only strengthen their role in immune health. Postbiotics represent a powerful blend of precision and reliability, positioning them as a key player in the future of immune health solutions.
FAQs
What makes postbiotics different from probiotics and prebiotics in supporting immune health?
Postbiotics stand apart from probiotics and prebiotics in both their makeup and the way they support the immune system. While probiotics are live microorganisms, postbiotics are non-living components or byproducts of microbes, such as short-chain fatty acids and cell fragments. These elements interact directly with immune cells, helping to regulate inflammation, strengthen the gut barrier, and maintain immune balance - all without requiring live bacteria to settle in the gut.
Prebiotics, in contrast, are non-digestible fibers that nourish the beneficial bacteria already living in the gut. While prebiotics indirectly support immune health by encouraging the growth of these microbes, postbiotics take a more direct approach. They deliver active compounds straight to the body, offering a more precise and predictable way to boost immune function. Plus, because postbiotics are non-living, they can be a safer option for people with weakened immune systems.
How can postbiotics benefit gut health and support the immune system?
Postbiotics are bioactive substances created during fermentation or as probiotics break down. Unlike probiotics, postbiotics are not alive, so they don’t need to colonize the gut to be effective. This makes them stable and easy to use. These compounds, which include short-chain fatty acids and bacteriocins, help fortify the gut barrier, maintain microbiome balance, and limit harmful bacteria - key factors in promoting gut health.
Beyond gut health, postbiotics also play a vital role in supporting the immune system. They can enhance anti-inflammatory responses, activate innate immune cells, and reduce oxidative stress. These effects are associated with a lower risk of chronic conditions, such as inflammatory bowel disease and metabolic syndrome. Products like Begin Rebirth RE-1™ combine postbiotics with prebiotics and probiotics, offering a well-rounded approach to rebalancing the microbiome and boosting immune health in as little as 7 days.
Why are postbiotics safe for people of all ages, including newborns and those with weakened immune systems?
Postbiotics are essentially inactive microbial cells, their byproducts, or components derived from these microorganisms that still maintain biological activity. Since they don’t contain live bacteria, they avoid the potential risks tied to probiotics, such as infections or bacterial migration. This makes them an excellent choice for vulnerable groups, including newborns and those with weakened immune systems.
Research indicates that postbiotics are generally well-tolerated across all age ranges, with very few reports of side effects. Their defined chemical makeup and absence of live microorganisms make them particularly safe for infants, older adults, and individuals with compromised immunity. Products like Begin Rebirth RE-1™ take advantage of these safety benefits, offering support for immune health and gut balance for everyone in the family - from the youngest to the oldest.