Prebiotics fuel the beneficial bacteria in your gut, influencing immune health and overall well-being. They work by producing short-chain fatty acids (SCFAs) that support immune balance, gut barrier integrity, and inflammation control. Research shows they can improve gut microbiota diversity, reduce inflammation markers, and even boost immunity in specific populations like the elderly or those with gut disorders.
Key takeaways:
- Prebiotics like inulin and FOS feed beneficial bacteria (Bifidobacterium, Lactobacillus), enhancing gut and immune health.
- Clinical trials reveal benefits like improved stool quality, reduced inflammation, and increased microbial diversity.
- Dosage and duration matter: effects range from 100 mg to 12 g daily, with sustained use recommended for lasting results.
- Combining prebiotics, probiotics, and postbiotics amplifies gut and immune benefits.
Emerging products, such as Begin Rebirth RE-1™, offer combined solutions for gut health, leveraging prebiotics, probiotics, and postbiotics for a multi-faceted approach. Consistent intake and tailored interventions hold promise for long-term gut-immune health.
Episode 15: A primer on prebiotics
How Prebiotics Change Gut Bacteria
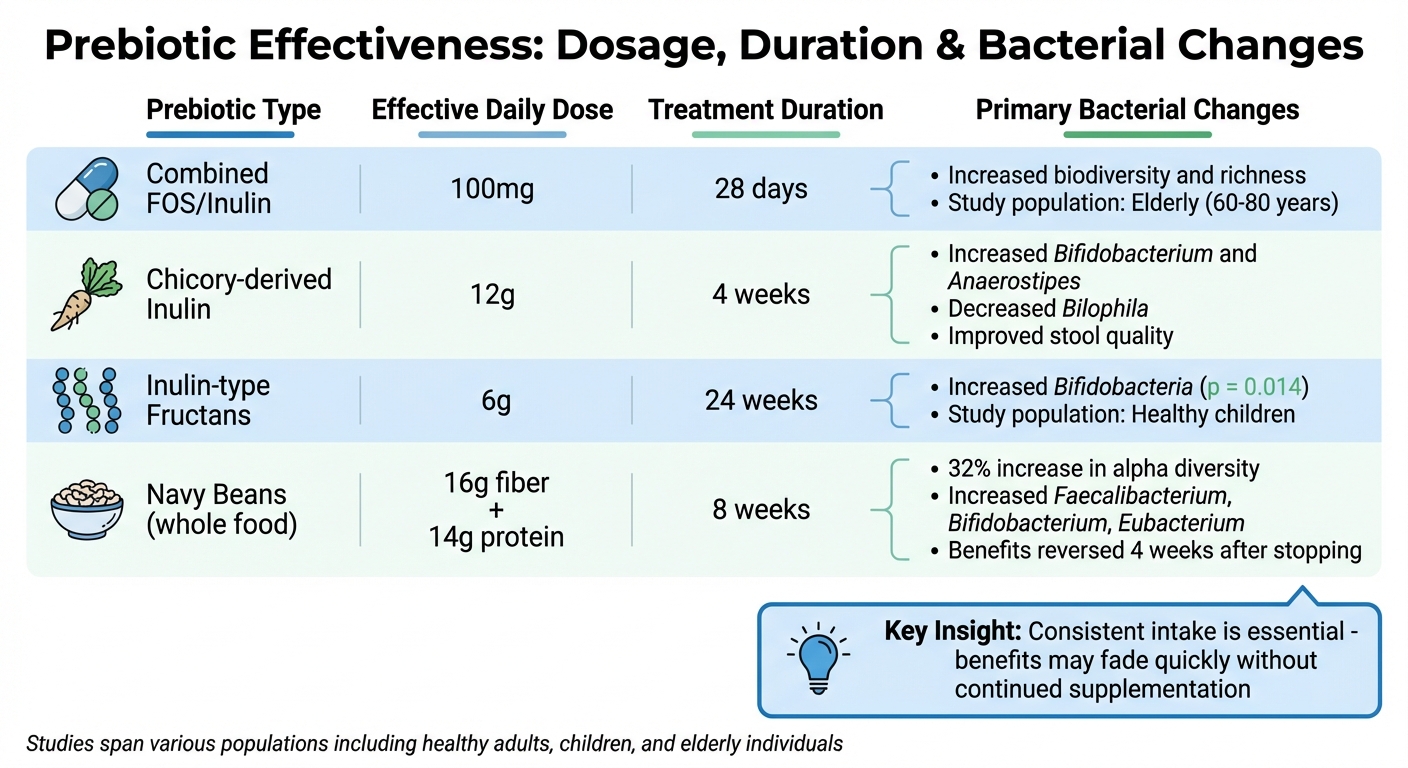
Prebiotic Dosage, Duration, and Bacterial Changes: Clinical Study Results
Prebiotics like inulin and fructo-oligosaccharides (FOS) take an interesting journey through the digestive system. They bypass digestion in the upper tract and reach the colon intact [7]. Here, they serve as a food source for beneficial bacteria such as Bifidobacterium and Lactobacillus, giving these helpful microbes an edge over harmful species.
Once fermented by these bacteria, prebiotic fibers are transformed into short-chain fatty acids (SCFAs). These SCFAs lower intestinal pH and produce natural antimicrobials like bacteriocins, hydrogen peroxide, and organic acids, which help to keep pathogens in check [10][6][5].
"The demonstrated selectivity of the effect of inulin allows customised interventions based on the nature of the dysbiosis targeted." - Jeroen Raes, Professor, Department of Microbiology and Immunology, KU Leuven [10]
Clinical Study Results on Bacterial Changes
Clinical research confirms how prebiotics can reshape gut bacteria. From 2010 to 2012, Professor Jeroen Raes led a study involving 42 healthy adults with mild constipation. Participants consumed 12g of chicory-derived Orafti inulin daily for four weeks. The results? Increased levels of Bifidobacterium and Anaerostipes (a butyrate producer) and a decrease in Bilophila, a bacterium linked to inflammation. The reduction in Bilophila coincided with softer stools and improved quality-of-life scores [10]. These shifts in gut bacteria also set the stage for immune benefits, which are explored later.
Another study, the "BE GONE" trial conducted by MD Anderson Cancer Center from July 2016 to January 2020, highlighted the impact of whole-food prebiotics. Dr. Carrie R. Daniel's team asked 48 obese participants with a history of colorectal neoplasia to consume one cup of pressure-cooked navy beans daily (providing 16g of fiber and 14g of protein) for eight weeks. This intervention led to a 32% increase in alpha diversity and significant growth in beneficial bacteria like Bifidobacterium, Faecalibacterium, and Eubacterium (p = 0.02). However, when participants stopped eating the beans, these benefits disappeared within four weeks [1].
In a 24-week study involving healthy children, a daily intake of 6g of inulin-type fructans (Orafti®) significantly boosted Bifidobacteria levels (p = 0.014) [11]. Similarly, a 2022 trial tested a synbiotic formulation combining 1×10⁹ CFU of L. acidophilus PBS066, L. plantarum PBS067, and B. animalis spp. lactis BL050 with 50mg each of FOS and inulin. Among 75 elderly participants (aged 60-80), this mix enhanced gut microbiota diversity and richness within 28 days, outperforming prebiotic-only and placebo groups [2][8].
Dosage and Treatment Duration
The effectiveness of prebiotics depends on dosage and how long they're taken. Studies show that daily doses can range from as little as 100mg to as much as 12g, depending on the specific prebiotic and the target group. For instance, in healthy adults, 100mg of combined FOS and inulin over 28 days was enough to positively influence gut bacteria [8]. On the other hand, 12g of chicory-derived inulin over four weeks significantly boosted stool frequency and selectively altered gut microbiota [10].
Treatment duration also plays a critical role. While some bacterial changes appear within four weeks, the BE GONE trial highlighted a challenge: microbiome improvements often fade quickly without continued supplementation. For long-term benefits, consistent intake is key. For example, studies on children have shown that sustained prebiotic use over 24 weeks can lead to lasting changes [10][11].
| Prebiotic Type | Effective Daily Dose | Treatment Duration | Primary Bacterial Changes |
|---|---|---|---|
| Combined FOS/Inulin | 100mg | 28 days | Increased biodiversity and richness [8] |
| Chicory-derived Inulin | 12g | 4 weeks | Increased Bifidobacterium and Anaerostipes; decreased Bilophila [10] |
| Inulin-type Fructans | 6g | 24 weeks | Increased Bifidobacteria (p = 0.014) [11] |
| Navy Beans (whole food) | 16g fiber + 14g protein | 8 weeks | 32% increase in alpha diversity; increased Faecalibacterium, Bifidobacterium, Eubacterium [1] |
Note: These studies span various populations, including healthy adults, children, and elderly individuals, reflecting the diversity of findings discussed in later sections.
Immune System Effects of Prebiotic Supplementation
Prebiotic supplementation brings about changes in the gut microbiota, which in turn directly impacts immune function. These shifts encourage the growth of beneficial bacteria that produce metabolites capable of influencing both local and systemic immune responses. The effects of these microbial changes are often measurable through specific immune markers.
Changes in Immune Markers
Clinical studies have demonstrated how prebiotic supplementation alters immune markers. For instance, a randomized, double-blind trial conducted in 2022 by researchers at the University of Milano-Bicocca examined the effects of daily supplementation with 100 mg of combined fructooligosaccharides (FOS) and inulin over 28 days in 75 elderly participants aged 60 to 80. The findings revealed changes in fecal β-defensin2 - an antimicrobial peptide - and calprotectin levels, along with a reduction in symptoms of common infectious diseases compared to a placebo group [8]. These results suggest that prebiotics enhance the gut's innate immune defenses and also modulate respiratory immunity through increased salivary IgA levels.
One key marker, fecal calprotectin, which signals intestinal inflammation, consistently decreases with prebiotic use. A randomized, placebo-controlled trial (NCT03852069) completed in February 2019 involved 24 obese participants who received either 16 g of native inulin or a maltodextrin placebo daily for three months. The prebiotic group showed a significant reduction in fecal calprotectin levels and an increase in rumenic acid, which is associated with the growth of Bifidobacterium (p = 0.031) [9]. Normal calprotectin levels are below 45 µg/g, and prebiotic interventions have been shown to help more individuals reach this threshold.
Another study, the BE GONE trial conducted at MD Anderson Cancer Center (completed in January 2020), explored broader immune effects. Forty-eight obese individuals with a history of colorectal neoplasia added one cup of organic navy beans to their diet daily for eight weeks. Researchers observed shifts in immune-regulating markers, including an increase in fibroblast-growth factor-19 (FGF-19) and a decrease in interleukin-10 receptor-α (IL-10Rα) (p = 0.01). These findings suggest that prebiotic supplementation can influence both metabolic and inflammatory pathways [1].
Biological Pathways
The immune-related changes observed with prebiotic use are underpinned by specific biological mechanisms. Short-chain fatty acids (SCFAs), particularly butyrate, play a key role in mediating communication between gut bacteria and immune cells. Alessandra De Giani and her team at the University of Milano-Bicocca explain:
"Prebiotic fibers are conventionally known to serve as substrate for probiotic commensal bacteria that release short-chain fatty acids in the intestinal tract... they then act on the local as well as the systemic immune cells and the gut-associated epithelial cells, primarily through G-protein-coupled receptor-mediated pathways" [3].
Butyrate, in particular, helps regulate immune responses by inhibiting HDAC-6 and -9 enzymes, which increases FOXP3 acetylation and promotes the proliferation of Treg cells - immune cells that suppress excessive inflammation [3][5]. Muhammad Bilal and colleagues at McGill University further emphasize:
"High intake of dietary fiber promotes microbial population producing short chain fatty acids (SCFA) that activate Treg cells through G protein coupled receptor 43 (GPR43) receptors to suppress inflammation" [5].
In addition to SCFA signaling, prebiotics also reinforce the gut barrier. They stimulate the production of tight junction proteins like zonula occludens-1 and mucin (MUC2), which reduce the movement of pro-inflammatory lipopolysaccharides (LPS) from the gut into the bloodstream [5][7]. Some prebiotics even interact directly with Toll-like receptors (TLRs) on innate immune cells, influencing cytokine secretion without requiring bacterial fermentation [3]. This combination of mechanisms explains how prebiotics can simultaneously affect gut health and systemic immune function.
sbb-itb-1bbfe7f
Clinical Evidence in Specific Populations
Elderly and Immunocompromised Individuals
As we age, our levels of bifidobacteria - helpful gut bacteria - naturally decline, which can lead to chronic low-grade inflammation. Research suggests that specific prebiotic treatments can counteract these age-related changes. For instance, a double-blind, placebo-controlled crossover study involving 44 healthy elderly participants showed promising results. Over 10 weeks, participants consumed 5.5 g of a trans-galactooligosaccharide mixture (B-GOS) daily. The findings? A significant increase in bifidobacteria, enhanced natural killer cell activity, and a reduction in pro-inflammatory markers like IL-6 and TNF-α [12][13].
"B-GOS administration to healthy elderly persons resulted in positive effects on both the microflora composition and the immune response." - J. Vulevic, University of Reading [12]
Another study conducted in France (March 2017–March 2018) examined the effects of a synbiotic combining Bifidobacterium animalis lactis and 4.95 g of fructo-oligosaccharides (FOS) in 27 middle-aged individuals (average age 58). After just 30 days, participants in the synbiotic group experienced reduced plasma levels of pro-inflammatory cytokines (IL-6, IL-8, and IL-17a) and increased levels of the anti-inflammatory cytokine IL-10 [14].
For individuals recovering from antibiotics - a period often marked by temporary immune suppression - daily intake of 2.5 g of galactooligosaccharides (GOS) has been shown to help restore bifidobacteria levels and promote butyrate production, a key compound for gut health [12].
These findings highlight how prebiotics can support not only the elderly and those recovering from antibiotics but also individuals managing chronic gut-immune conditions.
Gut-Immune Disorders and Chronic Inflammation
Prebiotics are also proving to be valuable in addressing chronic gut inflammation and rebalancing the microbiome in gut-immune disorders.
Patients with inflammatory bowel disease (IBD) often show reduced levels of butyrate-producing bacteria, such as Faecalibacterium prausnitzii and Roseburia hominis. These deficits are linked to more severe symptoms. Prebiotic supplementation has been shown to restore these beneficial microbes and strengthen the gut's protective barrier by enhancing the production of tight junction proteins like zonula occludens-1 [15].
In one 9-week study, patients with active ulcerative colitis (UC) who received high doses of oligofructose-enriched inulin experienced better clinical response and remission rates compared to those given lower doses [15]. Similarly, an 8-week trial using 1-kestose demonstrated higher remission rates in patients with mild-to-moderate UC compared to a placebo group [15].
Whole-food prebiotic approaches have also shown promise. For example, a study involving 41 adults who consumed 8 g/day of xylo-oligosaccharides (XOS) reported increased fecal bifidobacteria and reductions in specific immune markers [16].
These findings suggest that targeted prebiotic interventions could play a key role in managing chronic inflammatory conditions and improving overall gut health.
Combined Prebiotics, Probiotics, and Postbiotics
After looking at the individual effects of prebiotics and probiotics, let's dive into what happens when you combine them with postbiotics. Together, these three create a powerful synergy that supports gut health and strengthens the gut-immune connection more effectively than any one ingredient on its own [22].
Here’s how the combination works: Prebiotics act as fuel for beneficial bacteria, helping them thrive [17][18]. Probiotics, which are live microorganisms, compete with harmful bacteria for space and nutrients, while also producing helpful compounds. Postbiotics, on the other hand, are the byproducts of probiotic activity - such as short-chain fatty acids (SCFAs) - that deliver health benefits even when the probiotics themselves aren’t alive [17][18]. When probiotics break down prebiotics, they produce SCFAs, which help strengthen the gut lining and regulate immune responses [17][18].
"Pre-biotics, probiotics, and post-biotics are able to improve intestinal microbiota homeostasis, maintain gut barrier integrity, and modulate immune response." - Frontiers in Nutrition [4]
This combination also addresses a key challenge: if your gut struggles with fermentation, supplemental postbiotics can provide immediate benefits while prebiotics and probiotics work to restore balance [20][19]. Additionally, this trio boosts secretory IgA (sIgA) levels in the gut and saliva, enhancing protection against harmful microbes [17]. Some prebiotics even have a direct effect on immune pathways like MAPK and NF-κB, offering a dual layer of support [17]. Clinical studies back up this approach, showing how these components work together to improve gut-immune interactions.
For instance, a 16-week study involving obese children demonstrated the impact of this synergy. A synbiotic - a combination of prebiotics and probiotics - containing oligofructose-enriched inulin led to a 3.1% reduction in body weight z-scores and a 2.4% decrease in body fat percentage [21]. Postbiotics add another layer of advantage: they’re heat- and acid-resistant, making them stable and effective even in challenging environments like the stomach. They can also directly influence intestinal immune cells without relying on live bacteria [17].
Begin Rebirth RE-1™: A 3-in-1 Gut-Immune Formula
Building on this science, advanced products now combine the benefits of prebiotics, probiotics, and postbiotics into one formula. Begin Rebirth RE-1™ is a prime example, offering a 3-in-1 solution in a convenient daily sachet. This product includes prebiotics (4.5g of GOS and inulin), probiotics (500 billion CFU per serving with Human Origin Strains, or HOSt™), and postbiotics. The Lyosublime™ delivery system ensures these components survive the harsh stomach environment and reach the lower intestine, where they can interact with gut-associated lymphoid tissue [22].
The product is available in 7-day, 4-week, and 12-week reset programs, designed to quickly restore microbiome balance. Clinical research highlights that the origin of probiotic strains plays a crucial role in their ability to colonize the gut and interact with the immune system [22]. This principle is central to the HOSt™ technology in the formulation. Plus, it’s vegan, gluten-free, dairy-free, soy-free, and nut-free, and doesn’t require refrigeration.
This all-in-one approach offers both immediate and long-term benefits. Prebiotics fuel beneficial bacteria, probiotics help regulate immune responses and fight off harmful microbes, and postbiotics deliver anti-inflammatory effects right away. Together, they reinforce the gut barrier and limit the movement of toxins like lipopolysaccharide (LPS) into the bloodstream, which is a major factor in chronic inflammation [22][19].
Conclusion
Clinical studies highlight that prebiotics can significantly increase levels of Bifidobacterium and Lactobacillus while promoting the production of short-chain fatty acids (SCFAs) [23]. A systematic review of 22 randomized controlled trials confirmed that prebiotics deliver measurable health benefits by encouraging selective microbial fermentation [23]. These benefits include improved bowel regularity, strengthened gut barrier function, and reduced inflammation.
"Prebiotics are effective modulators of gut health, driving clinical benefits through selective microbial fermentation and SCFA production." - Nutrients Journal [23]
Prebiotics work in two main ways: they provide fuel for beneficial bacteria to produce SCFAs, and they directly engage innate immune cells via Toll-like receptors [3]. However, individual responses to prebiotics can vary widely, depending on the unique composition of each person's gut microbiota [23].
This growing body of evidence paves the way for personalized gut health solutions. Future research is focusing on tailoring interventions to fit individual microbiome profiles, aiming for more targeted and effective outcomes [23]. Additionally, there’s increasing interest in "3-in-1" formulations that combine prebiotics, probiotics, and postbiotics for a synergistic approach to gut and immune health [23][24]. Products like Begin Rebirth RE-1™ reflect this strategy, offering a clinically-supported formula designed to address the gut-immune connection through multiple mechanisms.
As we continue to explore the intricate relationship between the microbiome and the immune system, the emphasis will shift toward refining these interventions to meet individual needs. This includes investigating their broader effects, such as their influence on the gut-brain axis, opening up new possibilities for comprehensive health support [23].
FAQs
How do prebiotics support a healthy immune system?
Prebiotics play a crucial role in boosting the immune system by feeding beneficial gut bacteria like Bifidobacteria and Lactobacilli. These bacteria are essential for maintaining a healthy gut microbiome, which is tightly connected to immune function. By encouraging the growth of these helpful microbes, prebiotics strengthen the gut's natural defense system, helping it block harmful pathogens and manage inflammation.
When prebiotics are fermented in the gut, they produce short-chain fatty acids (SCFAs). These SCFAs not only support immune cells but also have anti-inflammatory properties, giving your body an extra layer of protection. A thriving gut microbiome, fueled by prebiotics, is key to keeping your immune system strong and your overall health in check.
What is the recommended daily dosage for prebiotics?
The amount of prebiotics you should take each day depends on the type and the specific health benefits you're aiming for. Common prebiotics like inulin, fructooligosaccharides (FOS), and galactooligosaccharides (GOS) are generally consumed in doses ranging from 3 to 10 grams daily to help with gut health and boost immune function. For example, a dose of about 5 grams per day is often enough to encourage the growth of beneficial bacteria like bifidobacteria. On the other hand, higher amounts - up to 10 grams daily - are sometimes recommended in clinical settings for added immune support.
When it comes to specialized prebiotics like human milk oligosaccharides (HMOs), the guidelines can vary, especially for infants, while recommendations for adults are less clear. A good approach is to start with a smaller dose and gradually increase it, depending on your body's tolerance and your specific health goals. To ensure you're taking the right amount, always check the product's instructions or consult with a healthcare professional.
Can prebiotics alone help improve chronic gut and immune health?
Prebiotics play a key role in supporting gut and immune health by feeding beneficial gut bacteria and encouraging the production of vital metabolites. Interestingly, research indicates that their effects on chronic gut-immune conditions improve significantly when paired with probiotics or synbiotics. Together, these elements work through various pathways to boost immune function and help restore balance in the gut.
For a more targeted solution, products like Begin Rebirth RE-1™ combine prebiotics, probiotics, and postbiotics in one formula. These specialized blends are crafted to work in harmony, offering quicker and more effective support for gut health and immune system balance.