Your gut and brain are in constant communication, and when that connection breaks down, it can lead to anxiety and depression. This relationship, known as the gut-brain axis, involves signals traveling between the central nervous system and the gut via nerves, hormones, and immune responses.
Key Takeaways:
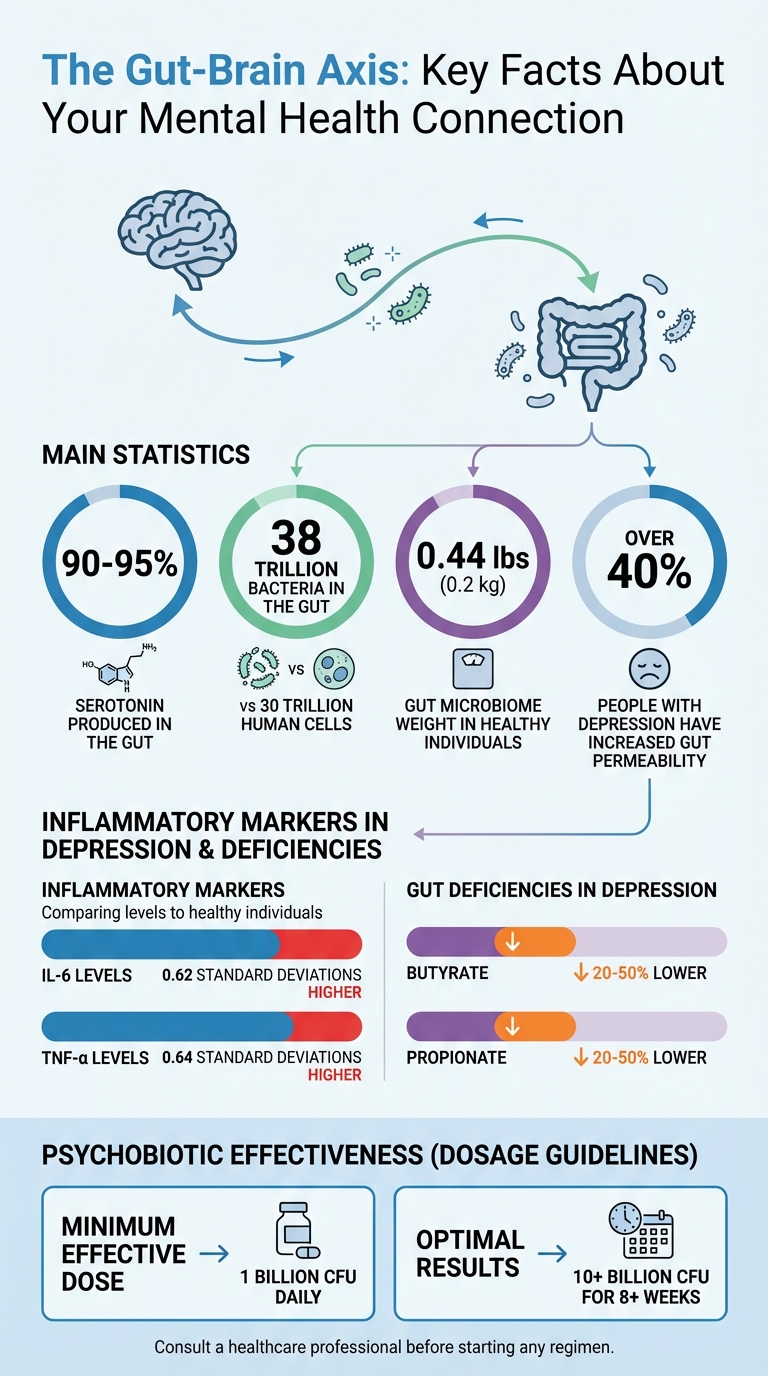
- 90% of serotonin, a mood-regulating neurotransmitter, is produced in the gut.
- An imbalance in gut bacteria (dysbiosis) can trigger inflammation and reduce neurotransmitter production, worsening mental health.
- Chronic stress, poor diet, and antibiotics can disrupt the gut microbiome, leading to "leaky gut" and neuroinflammation.
- Restoring balance with prebiotics, probiotics, and postbiotics supports gut health and may improve mood.
Research highlights how gut microbes influence neurotransmitters like serotonin, dopamine, and GABA, which regulate mood and stress. Addressing gut health through targeted interventions like psychobiotics and dietary changes offers a promising approach to managing anxiety and depression.
Gut-Brain Axis: Key Statistics on Mental Health and Microbiome Connection
Gut Dysbiosis and Mental Health
Understanding Gut Dysbiosis
Gut dysbiosis refers to an imbalance in the gut microbiome, which can mean fewer beneficial microbes or an overgrowth of harmful ones [9]. To put it into perspective, your gut contains around 38 trillion bacteria, compared to the 30 trillion cells that make up the human body [10]. Altogether, these microbes weigh about 0.44 pounds (0.2 kg) in a healthy individual [4]. When this balance is disrupted, it impacts not just digestion but also the body’s overall function.
There are many triggers for dysbiosis. Antibiotics, for instance, don’t discriminate - they kill both harmful and beneficial bacteria, lowering microbial diversity [9]. Chronic stress also plays a role, altering gut motility and immune responses, which reshapes the microbial community [10]. Stress hormones like norepinephrine and epinephrine can even encourage the growth of harmful bacteria [10][4]. Other factors include exposure to toxins, smoking, alcohol use, poor dietary habits (especially diets high in fat and sugar), and irregular sleep patterns [9][1].
This imbalance doesn’t just stay in the gut. It sets off a chain reaction involving the immune system and neurotransmitters, which can significantly affect mood and mental health.
How Dysbiosis Affects Anxiety and Depression
When gut bacteria are out of balance, it can trigger biological changes that contribute to anxiety and depression. One key issue is increased intestinal permeability, sometimes called "leaky gut." This allows bacterial toxins like lipopolysaccharides (LPS) to escape the gut and enter the bloodstream [1][3]. The immune system reacts by releasing pro-inflammatory cytokines (such as IL-1β, IL-6, and TNF-α), which can cross the blood-brain barrier and activate microglia, leading to neuroinflammation. This inflammation has been directly linked to symptoms of depression and anxiety [3][5].
Dysbiosis also interferes with neurotransmitter production. Normally, gut bacteria like Lactobacillus and Bifidobacterium help produce GABA and stimulate serotonin. But when harmful bacteria take over, this production drops [1][3]. Additionally, inflammation caused by dysbiosis diverts tryptophan - a key serotonin precursor - away from serotonin pathways and toward the kynurenine pathway, which produces neurotoxic compounds that negatively affect mood [1][3].
Studies have highlighted specific patterns in gut health and mental well-being. For example, beneficial bacteria like Faecalibacterium and Coprococcus, which produce butyrate, are consistently linked to improved quality of life [1]. When these microbes decline, mental health tends to worsen. In fact, research shows that over 40% of people with depression have increased gut permeability [4], illustrating how closely the gut-brain connection is tied to mental health.
| Factor | Impact on Gut Microbiota | Mental Health Consequence |
|---|---|---|
| Antibiotics | Reduces diversity and beneficial metabolites | Lower production of GABA and serotonin [9] |
| Chronic Stress | Increases gut permeability | LPS leakage leads to neuroinflammation [1][4] |
| High-Fat Diet | Promotes harmful bacterial growth | Linked to anxiety-like behaviors [5] |
| Sleep Disruption | Disrupts microbial metabolite rhythms | Weakens stress response and mood regulation [1] |
"Anxiety and depression are systemic conditions arising from integrated neural, immune, endocrine, metabolic, and circadian dysregulation, rather than isolated brain-based pathologies."
- Andrés López-Cortés, Cancer Research Group (CRG) [1]
Addressing gut dysbiosis means tackling these interconnected pathways - a topic we’ll dive into in the sections ahead.
How to Fix Your Brain-Gut Connection: Anxiety and the Brain-Gut Microbiome Axis
How the Gut and Brain Communicate
The gut and brain are constantly in conversation, exchanging information through various biological pathways. These include nerve networks, chemical signals, and hormonal systems that together influence mood, stress levels, and emotional health.
The Vagus Nerve Connection
The vagus nerve acts as the main communication line between the gut and brain. This nerve network, made up of sensory (afferent) and motor (efferent) fibers, sends signals back and forth nonstop [6].
Here’s how it works: the vagus nerve picks up chemical signals from the gut, such as short-chain fatty acids (SCFAs), bacterial byproducts like lipopolysaccharides (LPS), and neurotransmitters. It converts these signals into nerve impulses that travel to the brainstem’s nucleus tractus solitarius (NTS) before reaching areas like the amygdala and prefrontal cortex - key regions involved in emotional regulation and stress management [1][6].
The critical role of the vagus nerve is highlighted in studies where cutting it (a procedure called vagotomy) in animals eliminated the behavioral and neurochemical benefits of probiotics like Lactobacillus rhamnosus [11][1]. This underscores the vagus nerve’s importance in allowing gut microbes to impact mental health.
"The vagus nerve... is the major modulatory constitutive communication pathway between microbiota and the brain."
- Marilia Carabotti, Department of Internal Medicine and Medical Specialties, University Sapienza [11]
The vagus nerve also affects the hypothalamic-pituitary-adrenal (HPA) axis, helping to suppress stress hormones such as adrenocorticotropic hormone (ACTH) [10][6]. When the gut microbiome sends positive signals through the vagus nerve, it can help lower stress responses and improve emotional resilience. This connection allows gut bacteria to influence neurotransmitter production directly.
Gut Bacteria and Neurotransmitter Production
Gut bacteria play a remarkable role in producing neurotransmitters - the chemical messengers that regulate mood and cognitive function. For instance, they stimulate enterochromaffin cells in the gut to produce serotonin [12][6].
Different bacterial species are responsible for creating specific neurotransmitters. Lactobacillus and Bifidobacterium produce GABA, an inhibitory neurotransmitter that reduces anxiety and stress [1][12]. Similarly, Enterococcus, Escherichia, and Lactobacillus contribute to serotonin production, which is vital for mood regulation, sleep, and emotional balance [12][6]. Other species, like Bacillus, Lactobacillus, and Enterococcus, influence dopamine levels, which affect motivation, reward, and emotional states [12][6].
One standout example is Enterococcus faecium, which produces L-DOPA, a precursor to dopamine that can cross the blood-brain barrier and directly enhance dopamine levels in the brain [6]. While most neurotransmitters made by gut bacteria can’t cross this barrier, they still influence brain function by acting as precursors or by signaling through the vagus nerve [12][6].
| Neurotransmitter | Key Bacterial Producers | Role in Mental Health |
|---|---|---|
| GABA | Lactobacillus, Bifidobacterium | Reduces neuronal excitability, helping to ease anxiety [1][12] |
| Serotonin (5-HT) | Enterococcus, Escherichia, Lactobacillus | Regulates mood, sleep, and emotions; low levels linked to depression [12][6] |
| Dopamine (DA) | Bacillus, Lactobacillus, Enterococcus | Affects reward, motivation, and motor control; imbalances tied to anhedonia [12][6] |
When the gut microbiome becomes imbalanced (a condition known as dysbiosis), neurotransmitter production can drop significantly. Chronic inflammation, often associated with dysbiosis, diverts tryptophan (a serotonin precursor) away from serotonin synthesis and into the kynurenine pathway [1][7]. This shift not only lowers serotonin levels but also leads to the creation of neurotoxic compounds, contributing to what researchers call "serotonergic tone" depletion [1][13].
"The gut microbiota provides a novel way to alter neurotransmitter modulation in the brain and treat gut–brain axis diseases, such as anxiety and depression."
These microbial signals also interact with stress-regulating systems like the HPA axis.
The HPA Axis and Stress Response
Beyond neurotransmitter production, the gut influences the HPA axis, which governs the body’s stress response. When a stressor arises, the hypothalamus releases corticotropin-releasing hormone (CRH), prompting the pituitary gland to secrete ACTH. This, in turn, signals the adrenal glands to produce cortisol, the primary stress hormone. Normally, cortisol provides negative feedback, signaling the brain to reduce CRH and ACTH production once the stressor passes.
Gut dysbiosis can disrupt this system. Research shows that people with major depressive disorder have elevated levels of inflammatory markers like IL-6 (0.62 standard deviations higher) and TNF-α (0.64 standard deviations higher) compared to healthy individuals [13]. These inflammatory signals can overstimulate the HPA axis, leading to chronically high cortisol levels. Over time, this can damage brain areas like the hippocampus and prefrontal cortex, which are crucial for mood and stress regulation, creating a vicious cycle of stress and inflammation.
Experiments with germ-free animals - those raised without gut bacteria - show that they have exaggerated stress responses, with higher ACTH and cortisol levels during stress [11][1][5]. However, introducing beneficial bacteria like Bifidobacterium infantis can normalize their stress responses. This demonstrates how gut bacteria help regulate the stress response system.
Beneficial bacteria also produce short-chain fatty acids like butyrate, which improve glucocorticoid receptor sensitivity and reduce neuroinflammation. Studies in humans reveal that individuals with depression have 20% to 50% lower levels of butyrate and propionate compared to healthy controls [13]. This deficiency may weaken the blood-brain barrier and increase sensitivity to stress, making it harder for the body to manage stress effectively.
sbb-itb-1bbfe7f
Restoring Gut-Brain Balance
When it comes to improving mental health, tackling gut imbalance offers a promising path. The 3-in-1 eubiotic approach combines prebiotics (nutrients that feed good bacteria), probiotics (live beneficial microbes), and postbiotics (beneficial byproducts like short-chain fatty acids) to restore harmony in the gut-brain connection [7]. This method helps rebuild microbial diversity, strengthens the gut lining, and regulates neurotransmitter production.
The 3-in-1 Eubiotic Approach
Prebiotics, such as fructo-oligosaccharides (FOS), galacto-oligosaccharides (GOS), and inulin, act as food for beneficial bacteria, promoting the diversity often lacking in individuals with depression [15]. Probiotics, including strains from the Lactobacillus and Bifidobacterium families, can directly produce key neurotransmitters like GABA, serotonin, and dopamine - essential for emotional well-being [14]. Meanwhile, postbiotics like butyrate and acetate cross the blood-brain barrier, reducing neuroinflammation and supporting brain health [1][16].
Together, these components repair the gut lining, reduce harmful LPS leakage, and stabilize the HPA axis. Research indicates that psychobiotics are most effective at doses of at least 1 billion CFU daily, with doses exceeding 10 billion CFU over a minimum of 8 weeks showing optimal results [7]. Begin Rebirth RE-1™ delivers 500 billion CFU per serving in a clinically supported 3-in-1 eubiotic blend, featuring 4.5 grams of GOS and inulin, Human Origin Strains probiotics, and postbiotics, all optimized for absorption through its Lyosublime™ delivery system.
"The gut–brain axis has emerged as a transformative framework for understanding these disorders, emphasizing bidirectional communication between the central nervous system, the enteric nervous system, the endocrine and immune systems, and the gut microbiota."
- Jhommara Bautista, Frontiers in Psychiatry [1]
By restoring these functions, the gut barrier is repaired, paving the way for better neurotransmitter regulation.
Supporting Serotonin and Dopamine Pathways
Once the gut barrier is repaired, the next step is enhancing neurotransmitter production. A 2022 clinical study found that patients who took the probiotic strain Bifidobacterium breve CCFM1025 experienced a noticeable increase in fecal levels of 5-hydroxytryptophan (5-HTP), a serotonin precursor. This was linked to reduced depressive symptoms and improved gut health [7]. These findings align with the broader mechanisms of gut-brain communication by directly increasing neurotransmitter precursors.
Other strains, such as Lactobacillus helveticus R0052 and Bifidobacterium longum R0175, have been shown to lower cortisol levels and reduce psychological stress [1][15]. Similarly, Lactobacillus plantarum WLPL04 has demonstrated potential in alleviating anxiety-like behaviors by enhancing gut-brain signaling [15].
"The gut microbiota provides a novel way to alter neurotransmitter modulation in the brain and treat gut–brain axis diseases, such as anxiety and depression."
- Fei Huang, Frontiers in Cell and Developmental Biology [12]
Reducing Inflammation and Immune Dysfunction
Balancing the gut microbiome also helps curb inflammation and regulate immune function. People with major depressive disorder often show elevated inflammatory markers - IL-6 levels are 0.62 standard deviations higher, and TNF-α levels are 0.64 standard deviations higher than those in healthy individuals [13]. This inflammation can overstimulate the HPA axis, leading to persistently high cortisol levels and potential damage to brain areas that regulate mood.
Beneficial bacteria play a critical role here by producing butyrate, which inhibits HDAC activity, reduces neuroinflammation, and promotes neurotrophic factors [1][16]. Research suggests that individuals with depression may have 20% to 50% lower levels of butyrate and propionate compared to healthy controls [13]. Increasing these metabolites through fiber intake and probiotics strengthens the gut lining, limiting the movement of inflammatory compounds into the bloodstream. Additionally, this approach supports the growth of indole-producing bacteria, which help reduce microglial activation and ease anxiety-like behaviors [1].
Research Evidence and Future Developments
Clinical Studies on Gut Bacteria and Mood Disorders
Extensive research underscores the link between gut health and mental well-being. For instance, the Flemish Gut Flora Project revealed that butyrate-producing bacteria like Faecalibacterium and Coprococcus are tied to a higher quality of life, while their absence correlates with depression [1]. Similarly, individuals diagnosed with Major Depressive Disorder often show an increase in pro-inflammatory bacteria, such as Enterobacteriaceae and Klebsiella, while beneficial strains like Bacteroidetes are notably reduced [1][2].
Psychobiotics - probiotics aimed at improving mental health - are showing encouraging results. In a pilot study conducted in June 2023 at King’s College London, 49 participants with partial responses to antidepressants took a multi-strain probiotic for eight weeks. Those in the probiotic group reported significant reductions in both depression and anxiety symptoms compared to the placebo group [8]. A 2021 meta-analysis also supported the idea that psychobiotics are most effective when used alongside standard antidepressant treatments [8].
"For now, the best evidence supports psychobiotics as adjunctive therapies. Something that can complement, rather than replace, standard care."
- Wolfgang Marx, Nutritional Psychiatrist, Deakin University [8]
Another exciting development is Fecal Microbiota Transplantation (FMT), which holds potential for treatment-resistant depression. In early 2023, Dr. Valerie Taylor from the University of Calgary treated a patient with severe, treatment-resistant depression using a fecal microbiota transplant from a healthy donor. The patient experienced a significant reduction in symptoms. Reflecting on this approach, Dr. Taylor remarked:
"This is not how we'd thought about mental illness, as something that can be transferred the way you could catch measles." [8]
These findings are paving the way for more targeted and innovative treatments in mental health care.
Future Research Directions
The growing body of evidence is steering mental health research toward personalized interventions that leverage the gut microbiome. Researchers are now focusing on precision psychiatry, using multi-omics platforms - such as metagenomics, metabolomics, and meta-proteomics - to identify specific bacterial proteins and metabolites that directly influence brain function, including short-chain fatty acids [5][13]. This approach could help clinicians go beyond identifying bacterial presence to understanding their functional roles in the body.
Another area gaining traction is chrononutrition, which examines the gut–brain–circadian connection. Microbial communities in the gut appear to follow daily rhythms that align with stress responses [1]. Adjusting meal timing to sync with these natural cycles may enhance treatment outcomes. Additionally, advanced formulations like synbiotics and postbiotics are being developed to provide more consistent results compared to traditional probiotics [2][5].
Emerging technologies are also playing a key role. For instance, Magnetic Resonance Spectroscopy is being used to track changes in brain neurotransmitter levels - such as glutamate, GABA, and N-acetyl aspartate - in real time during probiotic treatments [5].
"Precision interventions, ranging from diet and psychobiotics to fecal microbiota transplantation, chrononutrition, and immune-modulatory strategies, offer promising avenues for personalized psychiatry."
- Andrés López-Cortés [1]
Conclusion
The gut–brain axis has reshaped how we think about mental health. Anxiety and depression are no longer seen as purely brain-based conditions. Instead, research highlights their connection to the trillions of microbes living in your gut. These microorganisms are responsible for producing about 90–95% of the body’s serotonin[12][17], directly influencing mental health through constant communication with the brain.
When gut dysbiosis occurs, the intestinal barrier weakens, allowing harmful endotoxins into the bloodstream. This triggers neuroinflammation and disrupts the chemicals that regulate mood. Studies consistently link butyrate-producing bacteria with better mental health, while their absence is associated with depression[1]. This evidence points to the importance of addressing gut health as part of mental health care.
"The gut–brain axis has emerged as a transformative framework for understanding these disorders, emphasizing bidirectional communication between the central nervous system, the enteric nervous system, the endocrine and immune systems, and the gut microbiota." - Frontiers in Psychiatry[1]
The good news? The microbiome can be adjusted, even in adulthood. Clinical research shows that targeted interventions - especially multi-strain psychobiotics combining prebiotics, probiotics, and postbiotics - can help restore microbial balance, lower inflammation, and support healthy neurotransmitter activity. Products like Begin Rebirth RE-1™, which delivers 500 billion CFU per serving with Human Origin Strains, offer a science-backed solution to rebalance the gut–brain connection and promote mental well-being.
FAQs
How does an imbalanced gut microbiome affect anxiety and depression?
An imbalanced gut microbiome, often referred to as gut dysbiosis, can have a profound effect on mental health. This is largely due to its disruption of the gut-brain axis - a complex communication system linking the gut and brain. When this balance is thrown off, it can trigger heightened stress responses, neuroinflammation, and alter the production of critical neurotransmitters like serotonin and dopamine, both of which play a key role in regulating mood.
Studies indicate that gut dysbiosis may amplify symptoms of anxiety and depression by interfering with these essential pathways. Keeping the gut microbiome in balance is not just important for digestion - it’s a cornerstone of emotional well-being. Addressing these imbalances could potentially ease mental health challenges and promote better overall emotional health.
How does the vagus nerve connect the gut and brain?
The vagus nerve serves as a critical bridge between your gut and brain, functioning as a two-way communication channel. Through this pathway, signals from gut microbes reach the central nervous system, allowing the gut microbiome to influence brain activity and even impact mood.
This connection is central to managing stress, regulating mood, and controlling inflammation, all of which are closely tied to mental health. Studies indicate that the vagus nerve plays a significant role in the microbiota-gut-brain axis, opening up possibilities for addressing issues like anxiety and depression by rebalancing this intricate system.
Can improving gut health with probiotics help with anxiety and depression?
Improving gut health with probiotics may contribute to managing anxiety and depression by supporting the gut-brain axis. Studies have shown that the gut microbiota plays an important role in regulating mood and emotional well-being. When the balance of gut bacteria is disrupted - a condition known as dysbiosis - it has been linked to mental health challenges like anxiety and depression.
Certain probiotic strains, such as Lactobacilli and Bifidobacteria, can help strengthen the gut-brain connection. They do this by encouraging a healthier gut microbiota, influencing the production of neurotransmitters, and reducing inflammation. Recent findings also indicate that combining probiotics with prebiotics and postbiotics may provide even greater benefits by restoring gut balance more effectively.
Although probiotics shouldn't replace traditional mental health treatments, they offer an additional way to support emotional well-being through gut health. It’s always a good idea to consult with a healthcare provider to find the most suitable approach for your specific needs.